|
The number proceeds in a linearly increasing fashion for the most part, once on the left of the table, and once on the right (see List of oxidation states of the elements), with some irregularities in the transition metals. The numbers indicate approximately the highest oxidation number of the elements in that group, and so indicate similar chemistry with other elements with the same numeral. Both use numerals ( Arabic or Roman) and letters A and B. Two earlier group number systems exist: CAS ( Chemical Abstracts Service) and old IUPAC. 
For example, group 16 is variously described as oxygen group and chalcogen. Also, groups may be identified by their topmost element or have a specific name. It replaces two older naming schemes that were mutually confusing. The modern numbering group 1 to group 18 is recommended by the International Union of Pure and Applied Chemistry (IUPAC). There are three systems of group numbering. The elements in a group have similar physical or chemical characteristics of the outermost electron shells of their atoms (i.e., the same core charge), as most chemical properties are dominated by the orbital location of the outermost electron. There are 18 numbered groups in the periodic table, but the f-block columns (between groups 2 and 3) are not numbered. 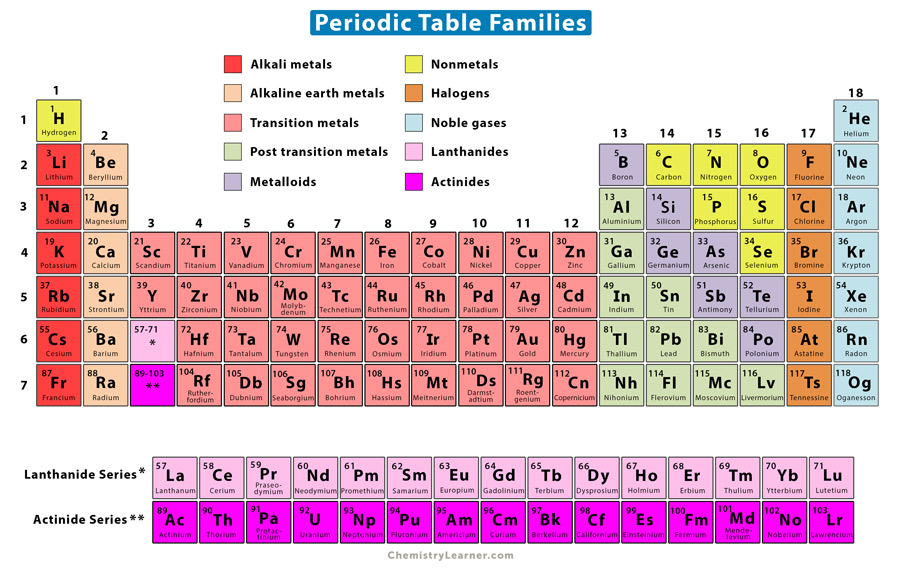
In chemistry, a group (also known as a family) is a column of elements in the periodic table of the chemical elements. Semimetals exhibit properties intermediate between those of metals and nonmetals.In the periodic table of the elements, each numbered column is a group. The elements can be broadly divided into metals, nonmetals, and semimetals. Some of the groups have widely-used common names, including the alkali metals (Group 1) and the alkaline earth metals (Group 2) on the far left, and the halogens (Group 17) and the noble gases (Group 18) on the far right. Elements that exhibit similar chemistry appear in vertical columns called groups (numbered 1–18 from left to right) the seven horizontal rows are called periods. It arranges of the elements in order of increasing atomic number. The periodic table is used as a predictive tool. As expected, semimetals exhibit properties intermediate between metals and nonmetals. 
Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. 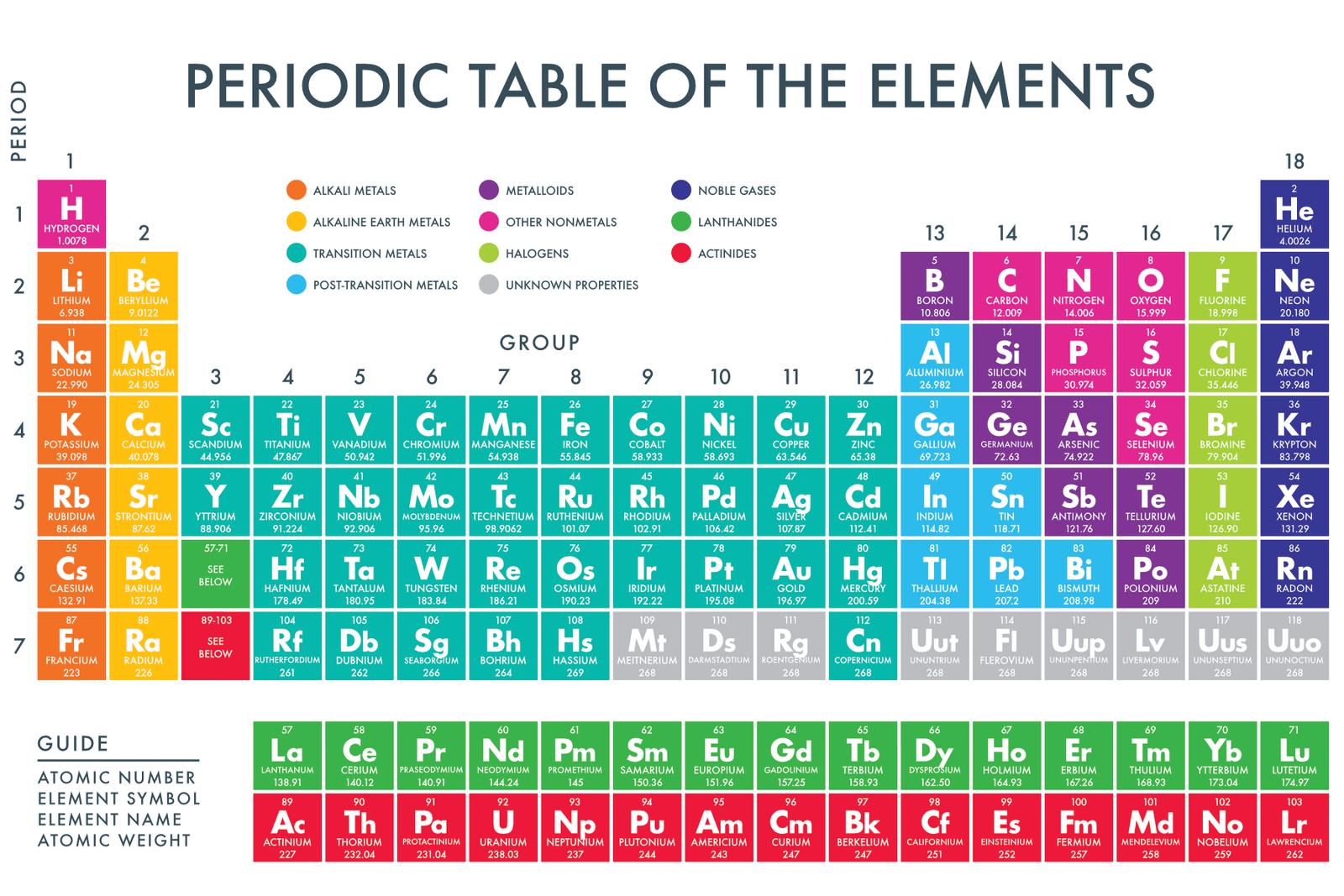
Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. The vast majority of the known elements are metals. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. The distinction between metals and nonmetals is one of the most fundamental in chemistry. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. \) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
